Peptide & Protein Nomenclature in Skincare
The goal of this educational post is to help you understand the difference between these two molecules, where they come from, and how to identify them on ingredient listings.
The simplest explanation as to what these molecules are is “a chain of amino acids”. If anyone remembers protein biochemistry from school, these are chains of amino acids, with varying chain lengths (number of amino acids), connected to each other via covalent amide/peptide bonds. How many amino acids are present in the chain, and their specific arrangement will determine if it is a peptide or protein, and its biochemical functionality. There are only 20 amino acids that are either naturally produced (non-essential) or required in diet (essential). From only these 20 amino acids there is an astonishing number of different molecules that can be created, each with completely different behavioral characteristics.
A peptide is any molecule that has anywhere from 2-50 amino acids in the chain (BTW in Biochemistry we call the amino acids “residues”). A molecule with 51 or more amino acid residues is called a protein. Peptides are often called small fragments of proteins. Most of the peptides you find in personal care products are synthetically created in the lab via chemical reactions that allow the amino acids to form these chains with high specificity. The scientists creating these molecules can look at larger proteins and assemble fractions of them in the lab. For instance, Matrixyl is the trademarked name for one of the oldest and most evidence-based peptides in skincare. It is known as a “matrikine”, which is essentially a fragment of larger matrix molecules (collagen, GAGs, et.). It tricks the skin into producing more of these substances.
Where do you get these peptides and proteins from? For the sake of simplicity, we will only be discussing lab-made and human cell-derived molecules. You can get peptides via chemical hydrolysis and protein denaturing of larger proteins “in nature” (i.e., human hair, animal hair, plants, etc.). Peptides or Proteins can be created in the lab, as mentioned, via a synthetic or recombinant process.
Synthetic is when you use no cellular material to make the molecule, just amino acids, and other chemicals and reagents to put the molecule together in the lab. Synthetics can be used to create completely new peptides that aren’t naturally found in the body or to create peptides/proteins that ARE found in the body. Recombinant is when you take human DNA coding for a specific protein, implant it in a non-human cell (oftentimes bacterial cells are used as the vector here, but plants cells have been used), hijack the cell’s genetic machinery, and get it to manufacture and release that exact same, molecularly identical human protein. With both methods, you are only producing ONE peptide or protein molecule at a time. And with synthetics, you are limited to relatively small molecules.
You can also get these molecules through the laboratory culture of human cells. You take the cells (yes, they must be alive) and in the lab you create the perfect environment for them to grow (mimicking their environment in the human body). They’ll start to grow and proliferate (produce copies of themselves), and as they do, they are in literal communication with each other by producing and pumping out their genetically pre-determined portfolio of molecules, which you will all recognize as being Cytokines, Growth Factors, etc. These are proteins, but the cells also produce a significant number of other biomolecules including peptides, chemokines, GAG’s, enzymes, lipids, sugars, etc. As you can see the cells aren’t producing one molecule at a time, they are producing a naturally secreted and physiologically balanced portfolio of these molecules. And they are produced in ABUNDANCE (depending on cell type)!
Ok, now that we know what these molecules are, and where they come from, how do you recognize them on an ingredient listing? This is where it can get slightly complicated, as indicated by someone’s recent post. Let’s begin by explaining the INCI. This stands for International Nomenclature for Cosmetic Ingredients. There is a global organization that determines the LEGAL way you are supposed to list your ingredients on your bottles/boxes, so that they can be identified and recognized globally.
In the INCI for a lot of these molecules you will often see “polypeptide”, or “oligopeptide”, of “tripeptide”, etc. What does it all mean? A peptide that consists of 2-20 amino acids in length is called an OLIGOPEPTIDE, and any chain that contains 20+ amino residues is called a POLYPEPTIDE (poly=many). However, the oligopeptide range can be further identified by how many amino acids are in the chain. EXAMPLE:
Dipeptide - 2 amino acids
Tripeptide - 3 amino acids
Tetrapeptide - 4 amino acids
Pentapeptide - 5 amino acids
Hexapeptide - 6 amino acids
Heptapeptide - 7 amino acids
Octapeptide - 8 amino acids
Nonapeptide - 9 amino acids
Decapeptide - 10 amino acids
And so on… USUALLY, if the peptide is in the 2-10 amino acid length range, it will be called by its residue content as explained above. Then from 10-20 amino acids, it is usually called Oligopeptide. Again, anything above this now becomes Polypeptide. Not to complicate things further, but there is an exception here! If the molecule consists of more than 20 amino acids, but there is only ONE single chain (not many linked together) it will still be called an Oligopeptide.
Let’s look at a few molecules and see how they would be listed (INCI) on the packaging. For isolated, individual molecules, you will often see the COMMON name listed with the INCI name.
EXAMPLE #1: Epidermal Growth Factor (EGF)
EGF would be the COMMON name, and the INCI name for this molecule is sh-Oligopeptide-1. Without this education, one would assume this is a peptide by category, because it says peptide in the name. But EGF is a protein because it has more than 50 amino acids. 53 to be specific, one of the smaller proteins. Remember I said there is an exception to the oligopeptide nomenclature? If EGF has 53 amino acids, how is it an Oligopeptide? Because EGF is made up ONE chain, not multiple chains pieced together.
You can see that the INCI name gives no indication as to how many amino acid residues there are. That would be far too complicated (unless less than 10 residues). So, what does the rest of the INCI name mean? The prefix says sh. That stands for synthetic human. This means that it was produced synthetically as mentioned above, but it is the exact same molecule you would find in the human body. Molecularly identical. If you see a prefix that says rh, which means recombinant human, it means it was produced in vector cells (bacteria usually) with human DNA. Either way, they would be identical molecules. You can find EGF in both rh and sh forms.
And finally, what does the 1 mean as the suffix? 99% of the time people think it has to do with number of amino acids, but that would be incorrect. It has to do with global INCI registration. The number indicates the order in which the molecule was registered. Think about it - you could have HUNDREDS or THOUSANDS of different peptides that all have the same tripeptide, oligopeptide, or polypeptide name. How do you tell them apart? The order in which they were registered! So, EGF is the very FIRST Oligopeptide to be registered in the global regulatory database. Hence its name is sh-Oligopeptide-1.
EXAMPLE #2: ARGIRELINE
This is the trademarked name for a peptide and its INCI name is Acetyl Hexapeptide-8. This is a synthetic peptide. It is a fragment of cellular membrane protein that the neurotoxin botulinum (Botox) binds to, hence it used to soften dynamic expression lines. What can you already tell me about the peptide? It is a Hexapeptide, so it has 6 amino acids! And it was the 8th Hexapeptide ever registered. What about the acetyl prefix?
MOST of these peptides (and certainly all proteins) are very large molecules. On average, each amino acid residue has a molar mass of around 110 Daltons. So, if we look at something like EGF, with 53 amino acids, its molar mass is around 6,000 Daltons. Argireline has a molar mass around 888 Daltons (well, about 660 Daltons prior to the acetylation explained below). We all know about the 500 Dalton Rule in skincare. There is more nuance to this, but largely anything over 500 Daltons will not penetrate the skin. So, what can you do to facilitate the penetration?
You can use penetration enhancers, various forms of liposomes, and/or you can add lipid moieties to the molecule. Say what? In Chemistry, a moiety is “a part of the molecule” that can be found in other molecules as well. These are often called functional groups, or sometimes “arms” when being colorful in explanation. They can be naturally occurring or, in this case, can be ADDED to the molecule to give it different biochemical properties. In the case of Argireline, the molecule has been acetylated, meaning one of the Hydrogens on the peptide has been replaced with an Acetyl functional group (CH3CO). This increases the molecules lipophilicity so it can wiggle its way into the skin’s lipid bilayers easier, even though it made the molecule slightly larger (888 Daltons). You might also see prefixes that say palmitoyl or myristoyl, and these are other “arms” added to the molecule to increase penetration into skin. In this case the arms are fatty acids. So, the molecule Acetyl Hexapeptide-8 is the 8th Hexapeptide ever registered, and it has been acetylated for enhanced skin penetration.
EXAMPLE #3: Human Cell Derived Proteins and Peptides
Now that we have looked at two different types of ISOLATED peptides and proteins, let’s explore how we get them from actual human cells. You will know you have human cell-derived molecules when you see the INCI Conditioned Media, and a prefix that tells you which cell was used, i.e., Human Bone Marrow Stem Cell Conditioned Media, Human Fibroblast Conditioned Media, or Human Adipose Stem Cell Conditioned Media.
What does this mean? The process is relatively straightforward. You take human cells (again, must be alive), and in the lab you put them in a cell culture dish (think petri dish) that has a special liquid inside. This liquid is called the media, and it consists of various nutrients, sugars, amino’s, electrolytes, etc. that the cells feed on to maintain their viability. We need to be sure that proper pH levels are maintained, osmolality is maintained, etc., so the cells can feel like they are at home. A few extra steps can be taken to ensure you are creating the perfect environment for the cells, such as anchoring them on tiny beads so they grow in a three-dimensional suspension and putting them in bioreactors to induce hypoxia (low O2 levels), though not all cells should be grown in hypoxic environments as it can stress them out and they become extra defensive (i.e., they produce a lot of pro-inflammatory molecules).
Now that you have created the perfect environment for them, they will start to grow, proliferate, and replicate themselves in the culture dish. As they do this, the cells will communicate with each other by releasing their portfolio of biomolecules such as Cytokines, Growth Factors, etc. These proteins are signaling molecules (think text messages between cells) that coordinate the activity of the cells. The molecules are released into the media. Once the cells have no more room to grow (we call this max expansion), fine filters are used to remove the whole living cells from the media so they can be transferred to another culture dish filled with media (we call this passage) to continue the process repeatedly (until the cells finally poop out, differentiate, become senescent, etc.). Once the cells have been removed, all you are left with is the media that is now rich in the proteins that were secreted into it. That media is now called conditioned.
As mentioned earlier, the cells will pump out all the molecules they are genetically programmed to produce. Cytokines, Growth Factors, Chemokines, Sugars, GAG’s, Enzymes, Peptides, etc. They do NOT produce one molecule at a time like the synthetic or recombinant processes. They are producing hundreds if not thousands of these molecules, which is why you will never see all the molecules they release listed on the ingredient deck. It would be far too complicated and LONG, requiring the largest packaging imaginable for a simple bottle. So, the global regulatory body decided that to standardize “the collection of all cell-derived molecules” for INCI purposes, they simply call it Conditioned Media, with the cell that was used as the prefix. Just know that within that one INCI, an immense number of molecules will be found within (though this is also cell-dependent, as some produce less molecules than others).
The media is now ready to be put into skincare products. However, if you just use the media as-is, you will run into a few issues. Stability and lack of skin penetration. Cell-derived molecules in a media need to be stabilized, and as mentioned above, most of the molecules are quite large so they can’t really get into the skin efficiently. One method used to stabilize and enhance delivery is to wrap the molecules inside liposomes, or a more advanced version of liposomes called Nanostructured Lipid Carriers. These small “lipid envelopes” protect and stabilize the precious molecules and promote penetration of them through human lipid bilayers and cell membranes.
SUMMARY
I hope this explanation can help you better identify the proteins and/or peptides that might be in your skincare products. We know that we can get these molecules from predominantly three sources:
· Completely synthetic where scientists are using building blocks (amino acids) and piecing them together to form chains (peptides). This can also be used to create human identical molecules and the peptide/protein prefix will be sh (synthetic human). This method has some limits and only peptides and small proteins can be created. Only 1 type of protein/peptide can be produced at a time.
· Recombinant processes where human DNA coded for the specific protein is implanted in non-human vector cells (bacterial), and the cell then takes those instructions and manufactures the human molecule that is identical to the ones found in the body. The prefix would be rh (recombinant human). Only 1 type of protein/peptide can be produced at a time. This is the most efficient and economical way to produce larger proteins at scale. In fact, the bulk of the insulin today is produced via recombinant technology. Insulin was the very first human protein drug produce via this tech.
· Human Cell Culture, where living cells are grown in the right environment to get them to pump out their full portfolio of the molecules, they are genetically hamstrung to produce. You get MYRIAD molecules, not just 1 like the two other methods. The INCI would read Conditioned Media with the prefix being the cell source that was used, i.e., Human Bone Marrow Stem Cell Conditioned Media.
And remember that these molecules are rather large for the most part and need assistance getting into the skin. Liposomes, Nano Lipids, lipophilic moieties, penetration enhancers, etc. can all be used to facilitate this. However, some proteins are so large that no matter what, they would never get into the skin. Collagen is the perfect example! The triple helix bundle contains around 3000 amino acid residues. That means it has a molar mass of around 330, 000 Daltons. Other than providing some surface moisture retention, collagen in skincare is nonsensical.
Usually, the INCI will contain the COMMON name AND the legally required INCI name. For instance, EGF will often present on the INCI as sh-Oligopeptide-1 (EGF), or EGF (sh-Oligopeptide-1). The only LEGAL requirement is to list the INCI name. You would have to do a search to find out what the peptide or protein is if the manufacturer doesn’t call it out.
And finally, this is just the BASICS of peptide/protein nomenclature. You will find some peptides that don’t fit this convention and can get really complicated for you to understand. For instance, one peptide’s INCI name is Dipeptide Diaminobutyroyl Benzylamide Diacetate and another one is called Tetradecyl Aminobutyroylvalylaminobutyric Urea Trifluouroacetate. These are very complex molecules with multiple groups added and are not as simple as your basic proteins/peptides. But that story and explanation will be for another day. J
Molecular Madness: Uncovering the truth behind Low Molecular Weight Hyaluronic Acid
Is it possible that our industry, often fueled by marketing compelling stories rather than science-based facts, could have it completely wrong as it relates to Hyaluronic Acid, its spectrum of molecular weights, and the value it provides to skin? As a cosmetic chemist, and 20-year industry veteran, I have seen my fair share of ingredients come available to us, each with varying levels of evidence to support topical usage. However, barring a few comically absurd ingredients (I am looking at you, Snail Snot & Sheep Placenta), no ingredient comes as close to being as illogical in the anti-aging world as Low Molecular Weight Hyaluronic Acid (LMW-HA) does. It is very rare that I would use the term irrefutable in a scientific conversation, but when it comes to the overwhelmingly abundant evidence within the human physiology and biochemistry literature that elucidates the pro-inflammatory behavior of LMW-HA, the data is just that…irrefutable. Let’s start with the basics.
Hyaluronic Acid is the most ubiquitous glycosaminoglycan (GAG) in the skin, an important component of the extracellular matrix, surrounding and supporting cells within the tissue architecture. This sugar polymer consists of repeating disaccharides of D-glucuronic acid and N-acetyl-D-glucosamine, linked together via glycosidic bonds (1). Primarily produced by dermal fibroblasts, and to a lesser extent within the epidermal keratinocyte (2), HA is involved in tissue wound repair, immune surveillance, cell migration, as well as acting as a molecular chaperone for a family of proteins known as hyaldherins (3) (4). HA is most famously known as a humectant, with myriad hydroxyl groups on its chemical structure able to bind to water molecules, keeping the skin hydrated, lubricated, and plump.
As we age, the amount of HA synthesized in our skin starts to decline, leading to a disproportionate portfolio of GAGs secreted, as well as reduced proliferation of fibroblasts (5). This, of course, is one of the numerous reasons we experience cutaneous aging (6) (7). Naturally, our industry was inclined to find ways to supplement the HA we lose over time, and so cosmetic suppliers provided us with topical HA and promised miracles would occur, touting it as an anti-aging holy grail. The truth, however, is much less exciting than that. Topically applied HA is far too large of a molecule to penetrate an intact Stratum Corneum, though interesting evidence does exist showing cutaneous HA penetration independent of molecular mass (8) (9). The value that topical HA provides to the skin is really two-fold – water retention, keeping the surface of your skin hydrated and happy, as well as acting as a pseudo-barrier, supporting the repair of a damaged Stratum Corneum due to a variety of external (and internal) aggressors.
Understanding the limitations topical HA has regarding replenishment of the naturally produced HA in the skin, and its decline over time, the “clever” (or not so clever) industry chemists decided to fragment the large HA polymer (HMW-HA) into smaller pieces (LMW-HA), to facilitate better delivery into the skin. This is where the disconnect between the scientific evidence and industry/brand marketers begins. To be fair, the molecular language in the literature is rather complex to understand, even for seasoned physicians. While the various molecular weights share some similarities as it relates to hydration, their biological functions in the skin are radically different.
Under normal, physiological conditions, the native HA that is synthesized in your skin is always of a high molecular weight (>1,000 kDa), supporting skin homeostasis and the stability of the skin architecture. The ONLY time you will find low molecular weight fragments of HA in the skin is subsequent to injury, and there is good reason for this. Upon injury, the amount of HA synthesis ramps up, as does frantic enzymatic activity (hyaluronidases). These enzymes are designed to catabolize the large HA polymer into smaller pieces, where they act as a biofeedback mechanism within the damaged tissue, triggering the alarm system of inflammation (10). Upon fragmentation, the low weight HA interacts with pattern-recognition receptors on cell surfaces, known as TLR-2 & TLR-4, or Toll-like Receptors. This receptor activation is the “on” switch for the built-in alarm system, with subsequent recruitment of pro-inflammatory cytokines, macrophages (11), and defensins (12) to the area of injury. This is the very definition of inflammation. While HMW-HA is known to attenuate the inflammatory response, LMW-HA is a potent trigger of inflammation and is known to promote fibrotic collagen deposition (13) (14). It is important to remember that when we discuss inflammation in this context, it does not mean the skin will be red, angry, and visibly inflamed. Sub-clinical inflammation underneath the skin can produce seemingly “positive” cosmetic benefits, since inflammation-mediated edema can make your skin plump (something these brands and suppliers touting LMW-HA are banking on).
So, while these small weight HA fragments do penetrate the skin, they are, unfortunately, a potent stimulus for inflammation. When we know definitively that inflammation is anathema to anti-aging, does it make any logical sense that exposing the skin to repeat inflammatory insults can somehow produce an opposite response? The answer is no, and the reams of evidence within the scientific literature support this (15) (16). There is no excuse here, and science will win over glorified sensationalism any day. In the words of my friend and mentor, world-renowned physician/researcher, Dr. Lance Setterfield, when it comes to the topical use of LMW-HA, it’s about time we “stop defending the indefensible”. I couldn’t agree more. *
*Originally published in the November 2021 issue of LNE Magazine
(1)
Weissmann B, Meyer K. The structure of hyalobiuronic acid and of hyaluronic acid from umbilical cord. J Am Chem Soc. 1954;76:1753–7.
(2)
Malaisse J, Pendaries V, Hontoir F, De Glas V, Van Vlaender D, Simon M, Lambert de Rouvroit C, Poumay Y, Flamion B. Hyaluronan Does Not Regulate Human Epidermal Keratinocyte Proliferation and Differentiation. J Biol Chem. 2016 Mar 18;291(12):6347-58.
(3)
Toole BP. Hyaluronan: from extracellular glue to pericellular cue. Nat Rev Cancer. 2004 Jul;4(7):528-39.
(4)
Dowthwaite GP, Edwards JCW, Pitsillides AA. An Essential Role for the Interaction Between Hyaluronan and Hyaluronan Binding Proteins During Joint Development. Journal of Histochemistry & Cytochemistry. 1998;46(5):641-651.
(5)
Matuoka K, Hasegawa N, Namba M, Smith GJ, Mitsui Y. A decrease in hyaluronic acid synthesis by aging human fibroblasts leading to heparan sulfate enrichment and growth reduction. Aging (Milano). 1989 Sep;1(1):47-54.
(6)
Tzellos TG, Klagas I, Vahtsevanos K, Triaridis S, Printza A, Kyrgidis A, Karakiulakis G, Zouboulis CC, Papakonstantinou E. Extrinsic ageing in the human skin is associated with alterations in the expression of hyaluronic acid and its metabolizing enzymes. Exp Dermatol. 2009 Dec;18(12):1028-35.
(7)
Meyer LJ, Stern R. Age-dependent changes of hyaluronan in human skin. J Invest Dermatol. 1994 Mar;102(3):385-9.
(8)
Essendoubi, Mohammed & Gobinet, Cyril & Reynaud, Romain & Angiboust, Jean-François & Manfait, M & Piot, Olivier. (2015). Human skin penetration of hyaluronic acid of different molecular weights as probed by Raman spectroscopy. Skin Research and Technology. 22. 10.1111/srt.12228.
(9)
Brown TJ, Alcorn D, Fraser JR. Absorption of hyaluronan applied to the surface of intact skin. J Invest Dermatol. 1999 Nov;113(5):740-6.
(10)
Krasiński R, Tchórzewski H. Hialuronian jako czynnik regulujacy proces zapalenia [Hyaluronan-mediated regulation of inflammation]. Postepy Hig Med Dosw (Online). 2007 Nov 19;61:683-9.
(11)
Rayahin JE, Buhrman JS, Zhang Y, Koh TJ, Gemeinhart RA. High and low molecular weight hyaluronic acid differentially influence macrophage activation. ACS Biomater Sci Eng. 2015 Jul 13;1(7):481-493.
(12)
Gariboldi S, Palazzo M, Zanobbio L, Selleri S, Sommariva M, Sfondrini L, Cavicchini S, Balsari A, Rumio C. Low molecular weight hyaluronic acid increases the self-defense of skin epithelium by induction of beta-defensin 2 via TLR2 and TLR4. J Immunol. 2008 Aug 1;181(3):2103-10.
(13)
Petrey AC, de la Motte CA. Hyaluronan, a crucial regulator of inflammation. Front Immunol. 2014 Mar 11;5:101.
(14)
Zgheib C, Xu J, Liechty KW. Targeting Inflammatory Cytokines and Extracellular Matrix Composition to Promote Wound Regeneration. Adv Wound Care (New Rochelle). 2014;3(4):344-355.
(15)
Ruppert SM, Hawn TR, Arrigoni A, Wight TN, Bollyky PL. Tissue integrity signals communicated by high-molecular weight hyaluronan and the resolution of inflammation. Immunol Res. 2014;58(2-3):186-192.
(16)
Papakonstantinou E, Roth M, Karakiulakis G. Hyaluronic acid: A key molecule in skin aging. Dermatoendocrinol. 2012;4(3):253-258.
Does Beta Glucan penetrate the skin?
Anyone that knows me understands that I’m such a stickler for accuracy and rationale when examining the foundational science of ingredients within the skincare ecosystem. I thoroughly evaluate any and all claims made by ingredient manufacturers, and critically analyze the evidence for scientific substantiation that validates (or invalidates) the claims. I lead with skepticism, as most in the scientific field should, as there’s much hocus-pocus to be found.
Beta Glucan is a good place to start. What is it? b-Glucan is a linear, glucose-only polysaccharide naturally found in the cell walls of yeast, fungi, oats, etc., with an average molecular weight WELL over 500 Daltons (closer to 500k Daltons). A very large polymer indeed! They are what’s known as biological response modifiers with immunomodulation properties. Topically, it’s claimed to have wound healing, collagen stimulating, and hydrating benefits. The humectant properties of b-Glucan are clear, given the multiple hydroxyl groups on the molecule. For the other biological properties to occur, this large polymer needs to penetrate the skin. So…does it? The industry wants you to believe it does, but what about the actual evidence?
I did an exhaustive search of the literature and found out that there’s only ONE study that everyone seems to cite, supporting the claim that b-Glucan penetrates the skin, independent of its massive size. The paper was filled with flaws and bias. First of all, the 2015 paper (Pillai, et al) was funded by Symrise, the manufacturer of the raw material. But what really struck me was the testing methodology, and had me scratching my head when reading the lab results. The experiment purports to document that the b-Glucan penetrates deeply into skin, but the protocol takes a skin biopsy and soaks it on a slide surface! This is NOT an intact barrier test they accurately reflects real world in-vivo conditions. The truth is you can get anything to penetrate devitalized epidermis on a slide! I consider this data worthless and a flimflam, yet it’s the one study everyone uses to support the claim.
The truth is, I think it is wise that this molecule doesn’t penetrate the skin. When you look at the evidence in the literature, the biological response seems defensive. The cutaneous immune system treat b-Glucan as antigen, and mounts an inflammatory response. This is evident by the polarization of the M1 macrophage phenotype. I plan to post more about this soon!
Welcome to the world of applied science in skincare! The more you know.
Is PRP worth it post-procedure? Elucidating the evidence
There has been much written about PRP (platelet rich plasma) and its use during microneedling (the Vampire Facial), with facial fillers (the Vampire Facelift), and other procedures. Briefly, PRP is prepared from the patient’s own blood in the physicians’ offices using specialized centrifugation and separation equipment. Regardless of what equipment is used (each varies in its efficiency of separating platelets), the objective is to exploit the high content of growth factors and cytokines found within the alpha granules contained in platelets. For their small size, alpha granules contain particularly high concentrations of these important cellular signaling molecules. And, to be fair, the use of PRP after “activation” of the alpha granules to release their bounty of bio-signals is well recognized as having proven medical benefit in a number of applications. These include orthopedics and sports medicine to promote healing and relieve pain e.g. bone defects, tendonitis, arthritis, ligament sprains and tears. It has also found usefulness in urogenital applications including the “O-shot”, “P-shot”, and penile augmentation. It is injected into the scalp with some success in restoring hair growth in balding patients. There is abundant literature attesting to these uses and others. PRP is the real deal, but not for everything. There are things for which it is less than ideal, and that includes aesthetics.
A little history: In the early 2000’s, the use of PRP extended into orthopedics to boost healing in bone grafts and fractures. Continued success encouraged its use in sports medicine for connective tissue repair. The first human study published by Mishra and Pavelko, associated with Stanford University, supported the use of PRP for chronic elbow tendinosis in 2006. This study reported a 60% improvement immediately, 81% at 6 months and 93% decrease in pain at the final two year follow up. For injuries where a little boost in inflammatory response to improve vascularity and increase oxygen delivery is good, PRP plays a positive role. Healing in an inflammatory environment promotes fibrosis, or scar tissue. In the context of orthopedics, you would rather have a fibrotic ligament than no ligament at all, right?
Aesthetic physicians jumped on the bandwagon a few years later. But does the evidence favor the use of PRP in aesthetics? Unfortunately, no.
If you inherently understand the fundamentals of aging you know that when it comes to anti-aging and facial aesthetics, the growing opinion is that inflammation is not desirable. In fact, it is something to be avoided whenever possible. That's why we are not fans of products containing pro-inflammatory ingredients such as fat stem cell conditioned media. Applying products on a daily basis that promote inflammation is not anti-aging, it's the opposite.
Although there are a great many different growth factors and cytokines contained within the alpha granules of platelets, there are five that predominate, regardless of the method of isolation. In the reference that follows, the five major bio-signals are identified as: VEGF (vascular endothelial growth factor), PDGF-BB (platelet derived growth factor), TGF-β1 (transforming growth factor beta-1), IGF-1 (insulin-like growth factor-1), bFGF (basic fibroblast growth factor). But that is just the beginning. There are many others of significance including: interleukins 1, 3, 4, 6, 8, 13 and 17; TGF-a, TGF- β2, TNF-a, IFN-a, EGF, aFGF and MMP (matrix metalloproteinases.) It is the sum of the collective effects of bio-signals that determines whether the pattern is pro-inflammatory or anti-inflammatory. The summary effect of PRP as depicted above is obvious: strongly inflammatory.
The effect then of applying PRP during microneedling is to exacerbate inflammation to jumpstart healing. Remember, inflammation when accentuated, persistent, or chronic is what leads to fibrosis (scarring) and excess pigmentation in susceptible individuals. As you can see, we're not fans of PRP although we do know it is popular (remember, the physician gets paid handsomely for using it.) Though PRP does initiate healing, it is not the type of healing our skin wants or needs. In fact, the data suggests PRP with MN for facial aesthetics actually adds nothing of value…so why bother?
THE TRUTH ABOUT PRP c/o Dr. Lawrence Koplin
Claim: PRP Facials are regenerative because your platelets accelerate healing and vitality.
The Truth: This statement is more marketing than biology. Cell survival and interaction with parent cells are scientifically relevant, but insufficiently understood elements of platelets in their function of healing and revitalizing tissue (2). There are no reliable studies showing that platelets are useful in healing tissue. PRP’s original function as a sports injury therapy has still not been proven effective. Doctors say they’re still not sure if it helps with chronic or acute injuries. Studies show that PRP may be more effective when compared to cortisol injections, but its results do not hold when compared to placebos.
Claim: More platelets means more healing.
The Truth: Dosage is critical with many medicines. Routinely, a higher dosage is worse for health. There is no evidence that increased platelets will speed up healing.
Claim: PRP is a natural treatment; it’s safe and healthy because there’s nothing foreign going into your body.
The Truth: There are lots of things in your body that are not beneficial to be extracted, increased in potency, and then returned to your system. Many hormones are part of the same healing process as platelets, but having too many will harm you.
Following the logic that having more of something natural is better, you would think that having more red blood cells is healthier, to give an example. Red blood cells are essential to life and provide vitality and healing. Yet hemochromatosis is a disease—an excess of iron—caused by the presence of too many red blood cells. In this case, absorbing too much is a problem, not a benefit.
On top of that, injecting materials into muscles is not unequivocally harmless. There’s conflicting evidence about PRP being myotoxic, meaning poisonous to muscles (3).
Skin infections can also occur between multiple PRP Facial sessions, with a higher risk posed to those with sensitive skin.
Claim: Studies have shown that PRP is effective.
The Truth: The only good news is coming from isolated or scientifically flawed studies. Reliable, randomized controlled trials are largely inconclusive. “The observed trend towards benefit with PRP use still remains questionable” (4). The first rigorous study testing the effectiveness of platelet injections finds they are no more effective than injecting saltwater (5).
1. Maria Del Russo, “The Cold, Bloody, (Kind of Disgusting) Truth About Vampire Facials,” http://www.refinery29.com/2016/10/128208/prp-treatment-vampire-facelift-review, October 31, 2016.
2. Vogel S M Gawaz, “Platelets in tissue repair: control of apoptosis and interactions with regenerative cells.” Blood 122, no. 15 (October 2013):2550–4. PubMed #23963043.
3. “Myotoxicity of Injections for Acute Muscle Injuries: A Systematic Review.” Sports Medicine 44, no. 7 (July 2014): 943-956, https://link.springer.com/article/10.1007/s40279-014-0186-6.
4. Dhillon RS, Schwarz EM, and Maloney MD, “Platelet-rich plasma therapy—future or trend?”, Arthritis Res Ther. 14, no. 4 (August 2012):219, https://www.ncbi.nlm.nih.gov/pubmed/22894643.
5. Gina Kolata, “Popular Blood Therapy May Not Work,” New York Times, (New York, NY), January 12, 2010.
Vet J. 2017 Jun;224:76-84. doi: 10.1016/j.tvjl.2017.04.005. Epub 2017 May 2.
Comparison of autologous bone marrow and adipose tissue-derived mesenchymal stem cells, and platelet-rich plasma, for treating surgically induced lesions of the equine superficial digital flexor tendon.
Abstract
Several therapies have been investigated for equine tendinopathies, but satisfactory long-term results have not been achieved consistently and a better understanding of the healing mechanism elicited by regenerative therapies is needed. The aim of this study was to assess the separate effects of autologous bone marrow (BM) and adipose tissue (AT) derived mesenchymal stem cells (MSCs), and platelet-rich plasma (PRP), for treating lesions induced in the superficial digital flexor tendon (SDFT) of horses. Lesions were created surgically in both SDFTs of the forelimbs of 12 horses and were treated with BM-MSCs (six tendons), AT-MSCs (six tendons), or PRP (six tendons). The remaining six tendons received lactated Ringer's solution as a control. Serial ultrasound assessment was performed prior to treatment and at 2, 6, 10, 20, and 45 weeks post-treatment. At 45 weeks, histopathology and gene expression analyses were performed. At week 6, the ultrasound echogenicity score in tendons treated with BM-MSCs suggested earlier improvement, whilst all treatment groups reached the same level at week 10, which was superior to the control group. Collagen orientation scores on histological examination suggested a better outcome in treated tendons. Gene expression was indicative of better tissue regeneration after all treatments, especially for BM-MSCs, as suggested by upregulation of collagen type I, decorin, tenascin, and matrix metalloproteinase III mRNA. Considering all findings, a clear beneficial effect was elicited by all treatments compared with the control group. Although differences between treatments were relatively small. BM-MSCs resulted in a better outcome than PRP and AT-MSCs.
J Dermatolog Treat. 2017 Aug 7:1-18. doi: 10.1080/09546634.2017.1365111. [Epub ahead of print]
Skin microneedling plus Platelet-Rich Plasma versus skin microneedling alone in the treatment of atrophic post-acne scars: a split-face comparative study.
Abstract
INTRODUCTION:
Acne scarring is a permanent disfiguring sequel, which can take varied morphological forms. Many therapeutic measures have been performed to improve acne scarring such as microneedling. Our objective is to evaluate the efficacy and safety of microneedling alone versus microneedling in combination with platelet rich plasma in the treatment of post-acne Methods: The study included 35 patients with mild to severe post-acne atrophic scar. All the patients received four sequential treatments of skin microneedling alone on the right side of the face and skin microneedling followed by topical application of PRP on the left side of the face with an interval of 3 weeks. Two blinded dermatologists evaluated the clinical response according to the qualitative global acne scarring system grading of Goodman & Baron. Patients are queried about their satisfaction with the treatment outcomes.
RESULTS:
The study included 35 patients with a mean age of 24.7±6.8 years. There was a significant improvement in the degree of scar severity before and after treatment on both sides. Regarding patient satisfaction grades there was a significant improvement after both treatment modalities with insignificant differences between both treatment modalities.
CONCLUSION:
Both microneedling and microneedling in combined with PRP showed satisfactory results. PRP adds NOTHING to microneedling...so why bother?
Am J Sports Med. 2012 Jun;40(6):1274-81. doi: 10.1177/0363546512442334. Epub 2012 Apr 10.
Comparison of the acute inflammatory response of two commercial platelet-rich plasma systems in healthy rabbit tendons.
Dragoo JL1, Braun HJ, Durham JL, Ridley BA, Odegaard JI, Luong R, Arnoczky SP.
Abstract
BACKGROUND:
Numerous studies have shown platelet-rich plasma (PRP) preparations differ with respect to the inclusion of certain blood components, which may affect the host's cellular response.
HYPOTHESIS:
This study evaluated the inflammatory effect of Biomet GPS III leukocyte-rich PRP (LR-PRP) versus MTF Cascade leukocyte-poor PRP (LP-PRP) after intratendinous injection in an animal model. The authors anticipated that LR-PRP would incite a greater acute inflammatory response than LP-PRP.
STUDY DESIGN:
Controlled laboratory study.
METHODS:
A total of 17 skeletally mature New Zealand White rabbits were tested. In all cases, healthy patellar tendons were treated. In the control animals, one patellar tendon was injected with 2 mL autologous whole blood, and the other was injected with 2 mL sterile saline. Seven total tendons were injected with whole blood, and 7 tendons were injected with saline. In the experimental animals, one patellar tendon was injected with 2 mL LR-PRP, and the other was injected with 2 mL LP-PRP. Ten tendons were injected with LR-PRP, and 10 tendons were injected with LP-PRP. Animals were euthanized at 5 or 14 days after injection. Tendons were harvested and stained using hematoxylin and eosin and scored semi-quantitatively for total white blood cells (WBCs), mononuclear cells (macrophages and lymphocytes), polymorphonuclear cells (PMNs), vascularity, fiber structure, and fibrosis.
RESULTS:
At 5 days after injection, tendons treated with LR-PRP had significantly greater overall tendon scores (6.3 ± 1.79 vs 1.8 ± 1.64, P = .012), as well as mean scores for fiber structure (1.4 ± 0.22 vs 0.50 ± 0.50, P = .012), denoting disrupted composition, total WBCs (1.1 ± 0.89 vs 0.10 ± 0.22, P = .014), mononuclear cells (macrophages and lymphocytes) (0.80 ± 0.45 vs 0.10 ± 0.22, P = .014), vascularity (1.7 ± 0.27 vs 0.80 ± 0.16, P = .008), and fibrosis (1.0 ± 0.35 vs 0.3 ± 0.45, P = .037) compared with tendons treated with LP-PRP. Otherwise, there were no significant differences in mononuclear cells (P = .590), PMN cells (P = 1.00), total WBCs (P = .811), vascularity (P = .650), or total tendon score (P = .596) in any of the treatment groups at 14 days.
CONCLUSION:
Compared with leukocyte-poor Cascade PRP, leukocyte-rich GPS III PRP causes a significantly greater acute inflammatory response at 5 days after injection. There is no significant difference in the inflammatory response or cellularity regardless of the injection type at 14 days after intratendinous injection.
CLINICAL RELEVANCE:
Platelet-rich plasma injections are frequently prepared using commercial systems and are administered for clinical treatment of chronic tendinopathy. It is important to characterize the cellular responses elucidated by different injection preparations to further understand their effect on tissue healing and aid clinical decision-making. Future investigations are necessary to apply these findings to the clinical setting. HIGHLY INFLAMMATORY
Arthroscopy. 2019 Oct;35(10):2885-2886. doi: 10.1016/j.arthro.2019.06.015.
Editorial Commentary: Platelet-Rich Plasma or Profit-Rich Placebo: Variability of Composition, Concentration, Preparation and Many Other Yet-Unknown Factors Determine Effectiveness.
Abstract
Platelet-rich plasma (PRP) frequently is used in orthopedics, and its application is supported by clinical studies for a variety of conditions. For knee osteoarthritis, it is more effective than hyaluronic acid, providing significantly better pain relief and functional improvement. However, different compositions of PRP, absolute platelet counts, and many other physiological and demographic variables will influence the effectiveness. Variability of the composition of the ingredients of PRP surely has a substantial influence on the outcome.
Even in orthopedics, the benefits are highly questioned!
The LARGEST meta-analysis of all the literature on PRP/aesthetics suggests no benefit.
J Dermatolog Treat. 2016;27(3):285-9. doi: 10.3109/09546634.2015.1094178. Epub 2015 Oct 14.
Applications of platelet-rich plasma in dermatology: A critical appraisal of the literature.
Author information
Abstract
Platelet-rich plasma (PRP) is an autologous blood-derived product enriched in platelets, growth factors, chemokines, and cytokines. Initial applications were predominantly in musculoskeletal and maxillofacial fields, however, in recent years, it has been used for a range of dermatological indications including wound healing, fat grafting, alopecia, scar revision, and dermal volume augmentation. Here, we critically appraise the literature relating to the usage of PRP within Dermatology. We have evaluated in vitro data, preclinical animal studies, and human trials. We conclude that whilst the literature may be consistent with a modest benefit for specific indications, there is not sufficient evidence supporting the efficacy of PRP to justify a role in routine dermatological practice at the present time. However, since PRP is generally well tolerated with few reported complications, further study may be justified in the context of organized trials.
CONCLUSION: PRP adds NOTHING beneficial to post-procedure skin.
Does Hyaluronic Acid actually "rob" your skin of moisture?
So, does Hyaluronic Acid applied topically pull water from your skin and “rob” it of its moisture? You will all be quite surprised that the answer to this is largely NO, with some caveats. Let me explain the science.
Molecules that draw water to themselves, or are hygroscopic, are known as humectants. From a chemistry perspective, humectants share similar properties in that they contain multiple hydroxyl groups (-OH), which can interact with water molecules via hydrogen bonding.
A variety of humectants are native to the skin such as glycerol, hyaluronic acid, urea, etc. In topical preparations, these humectants can help supplement the ones naturally present to increase water concentration to the tissue. Cutaneous hydration is paramount to its health and homeostasis, as many of the enzymatic and biochemical pathways that keep the skin functioning normally are hydrolytic, or water-dependent (the skin itself, and the body, contain roughly 65-70% water…more on this later).
So, the theory goes that if you apply these humectants to the skin in an atmosphere of increased humidity, they will pull the moisture (water) from the environment, and if applied in a dry climate they will pull the moisture (water) from the deeper layers of the skin, “robbing” it of moisture. The former part of this statement is factual, the latter part…not so much (again, with caveats).
Let’s look at glycerol, more commonly known as glycerin, which is by far the most effective and scientifically sound humectant for skin when applied topically (yeah, it kicks hyaluronic acid in the butt and says, “take it, bi*ch!”). When applied topically, glycerin and all those “thirsty” hydroxyl groups, are ready to attract the water molecules to themselves. Yes, it can pull the water from an environment with increased relative humidity, it can hold onto water that is sprayed on the skin before applying it, or it can attract water from the underlying tissue. In fact, the size of the glycerol molecule is quite small (92 g/mol, or 92 Daltons), that it can easily penetrate the skin.
The big question at hand is does this attraction of water from the deeper layers to the upper layers, “rob” our skin of moisture? Guess what, for most of us the answer is a resounding NO! What?! You say this is what you have been told by brands, educators, physicians, and beauty influencers your whole life?! I am in SHOCK that the industry would feed you inaccurate information…that NEVER happens! ;-P
How is this possible you ask? The simple answer…because our body, and our skin, are made up of 70% water. “Robbing” typically means that you are taking something valuable that often cannot be replaced. This is important in this context, and not just semantics. The in-depth answer? Water in the body/skin is not limited in supply. If we use humectants, and they draw deeper water stores to the upper levels of the skin, they are not robbing the water, they are simply redistributing it, and any water drawn out would be replaced via your circulation if there is water to replace it. This is called the concentration gradient (science…it kind of matters). Our skin is quite privileged in terms of water distribution in a total body “drought”. However, if you are clinically dehydrated, can longer sweat, and there is a limited supply of water in your body, then we are in a different situation and you are in dangerous territory. By the way, if this is you, please stop reading this and go to the hospital right away. You have far more to worry about than the ingredients in your skincare.
So, if the water being attracted by the humectant is coming from the deeper layers of the skin, and that water is readily replaced by water from our circulation, what IS the problem with humectants? This is where that concentration gradient can turn on us, especially in dry climates. You see, under normal physiological conditions, the relative humidity of the skin underneath our skin barrier (stratum corneum) is high, around 99%. However, the external environment, depending on where you live, has a relative humidity range between 40-60%. This is an extreme water gradient across the skin’s membrane. Concentration gradient is basically where, to keep things “balanced”, the amount of water inside a membrane, if it is higher than outside the membrane, will deliver water to the outside of the membrane to equalize the situation. This is not an even trade, however. A dry, low humidity environment does not give back. This is essentially what we mean by trans epidermal water loss (TEWL).
Some of you might say that I just contradicted myself. You will say that I just proved humectants can rob the skin of moisture. But I did not. The water supply is still there to replenish. The problem here is twofold. Firstly, I mentioned the “physiologically normal” conditions above. Let’s be honest, for most of us, there is nothing physiologically normal about our skin and its condition. What do I mean?
Let’s talk about how our skin DOES hold on to water under normal, physiological conditions. If your mind is thinking about the skin barrier (stratum corneum), you win the million-dollar prize. The skin barrier is formed when maturing epidermal keratinocytes lose their nucleus to become surface corneocytes. In addition, lamellar bodies inside the keratinocytes release lipids, that will then surround the corneocytes. This is our “waterproof” lipid barrier. Oh, by the way, the processes I just mentioned are largely water-dependent. Under these physiological conditions, water contained in the lower levels of the skin, or any aqueous material, cannot penetrate beyond this lipid-rich structure at the surface of our skin.
However, as mentioned, our skin does not live a physiologically normal life. Our barrier is constantly being compromised, either from the products we use, over-exfoliation, age, or the sun's UV rays that compromise the integrity of the barrier. These factors alone can contribute to water being released from our skin, and even more so in a dry climate due to that concentration gradient increasing water flux. This brings me to the second highlight of the issue…humectants should NEVER be used alone. Why attract all that water to the skin if you aren’t going to “seal it in”? As great as these humectants can be, what truly protects your skin and holds the water in is your lipid barrier. You should always be following up the application of a humectant (if a solo product) with a properly formulated barrier moisturizer, to help reestablish those normal, physiological conditions as much as possible. Some barrier moisturizers have the humectants built-in, and that is perfectly fine as well. At the end of the day, what determines the level of TEWL in our skin is the water concentration gradient between the inside of our skin and the outside environment and the integrity of the skin’s lipid barrier.
If you are still following, bear with me. This is where I get into the good stuff. Hyaluronic Acid (HA). I mean that is what I came here to talk about, right? Let me first tell you that HA is a bit of a different animal compared to what I described above, at least from the perspective of us applying it topically. As you all know, HA is another humectant that our skin naturally produces. It is also known as a glycosaminoglycan (GAG), and it’s the most ubiquitous humectant found in our skin that gives it buoyancy, plumpness, and cushion, among many other benefits that I won’t go into now (Stay focused, Daniel!). What makes topical HA different than the other humectants when applied topically? One simple word – SATURATION!
Most people (surprisingly) don’t realize that 100% PURE Hyaluronic Acid is a solid powder. This is never applied to your skin NEAT. It is always hydrated in water first, then mixed into a formula that eventually goes on the skin. Because HA is such an effective hygroscopic agent, usage levels in skincare are quite low. Most of the time HA is found at less than 1% in a topical product, with 0.2-0.5% being the average. Anything above 2% and the formula would be quite thick, especially if you are using high molecular weight (which you should, by the way). Those brands that claim they have 20%, 50%, 90%, HA in their products…THEY ARE LIARS! HA powder reconstituted in water, at these levels, would be as hard as a ROCK!
So, HA in pure form has not had any of its water-binding sites activated yet. However, when you mix it in water, those sites become hydrated/saturated, and then that water-saturated HA gets applied to the skin. Are you listening? When you apply HA to your skin in any cosmetic formula, it has ALREADY been saturated with water molecules, meaning it is no longer attracting water to itself! It is not “thirsty” anymore. Let me break down some chemistry and math to help you understand:
HA is a sugar polymer, and a pretty large polymer at that, with smaller disaccharide units on its structure. Native HA that your skin releases are high molecular weight polymers. This means many polymers linked together in a large chain. Within these chains are water binding sites, just like any humectant. Each water-binding site on the HA polymer can hold about 15 water molecules. Let’s look at the amount of water binding sites on a large, high molecular weight polymer of HA used in skincare. I will use 2,000 kDa, or 2M Daltons as an example. I have done the math for you, and an HA polymer of this size would have 5,114 units that can bind to water molecules. If each unit can bind to 15 water molecules, the 2 Million Dalton HA polymer can attach to 76,710 water molecules before saturation.
So, how many H2O molecules are inside water? Let’s look at a formula of pure HA saturated in water. Let’s use 1% HA and make a 100-gram sample. In this case, 1% = 1 gram. If we have 1 gram of HA, we have 99 grams of water. How many H2O molecules are in 1 gram of water? A LOT! 1 gram of water contains 3.3455 x 10^22 water molecules. I know, I know. I will do the math for you again. The latter part of that equation is 10 to the 22nd power. Are you ready for it? That number is 10 SEXTILLION! I will take that in dollars, thank you. Now times that by 3.3455…and you have more water molecules than you can even imagine. If that formula has 99 grams of water to its 1 gram of HA….I don’t think I need to do more math to get you to see my point.
The HA you apply to your skin is FULLY SATURATED. There is some theory that even when fully saturated, HA can bind to more water, due to the general chemistry principles we know of about water, and how each water molecule can bind to 4 others, without needing any other influence. This is based on theory and has not been proven in the literature for HA. Even so, given my breakdown above, there are still more than enough water molecules in that 99 grams of water for this to still leave water molecules behind, and the HA no longer “seeking” it from somewhere else (i.e., your lower skin levels).
Next, you will tell me “But I FEEL dry after I apply my HA!”. Let me explain! Again, if you are using HA in your skincare, it should always be high molecular weight. When you apply that HA to your skin and the excess water evaporates, it will leave the polymer film on the skin which can make the skin feel dry/tight, but it is STILL acting as a water-loss barrier, keeping the underlying skin happy. I addition, HA can spontaneously cross-link, creating a stronger mesh-like network on the skin, which will also lead to that temporary tightening effect. High Weight HA is less of a humectant than it is an occlusive, believe it or not. That is its real value. It will hold on to the saturated water molecules and create a barrier that helps prevent TEWL. High Weight HA is too big of a polymer to enter the skin.
However, HA, just like all in this class of humectants, should never be used alone. In fact, HA is the worst offender in this context. It is NOT an elegant molecule in a formula by itself. Have you ever looked at all these popular HA products on the market? They are loaded with other materials that give the skin some comfort (glycerin, possibly lipids, etc.) A pure, 1% HA serum, with nothing else but water in it, will not feel great on the skin if used alone, especially if it’s HIGH WEIGHT HA, which it should be. Why bring all these water molecules to the skin if you are not going to seal them in?
What about Low Molecular Weight HA? While yes, this does have the ability to penetrate better into the skin and can maintain a bit more humectant properties compared to High Weight, the same rules apply as far as water is able to be replenished. The problem with Low Molecular Weight HA is that it is INFLAMMATORY. I can take up a whole article discussing this, so I won’t get into it here. Just know this is IRREFUTABLE. LMW-HA can increase TEWL because the inflammatory cascade it triggers can weaken the skin barrier, allowing water to evaporate. LMW-HA is bad news, no matter how you look at it.
So, to sum things up, HA used topically is fully saturated with water. High Molecular Weight HA, if used alone, will leave a large polymeric film on the surface that can FEEL tight/dry, but is not robbing or pulling moisture from your skin. Still, seal it up! Low Weight HA…yeah, throw it in the garbage. And all other humectants…use in a responsible way, with a balanced portfolio of products that restore barrier function and hold that water in. Make sense?
Is this truly nature's retinol?
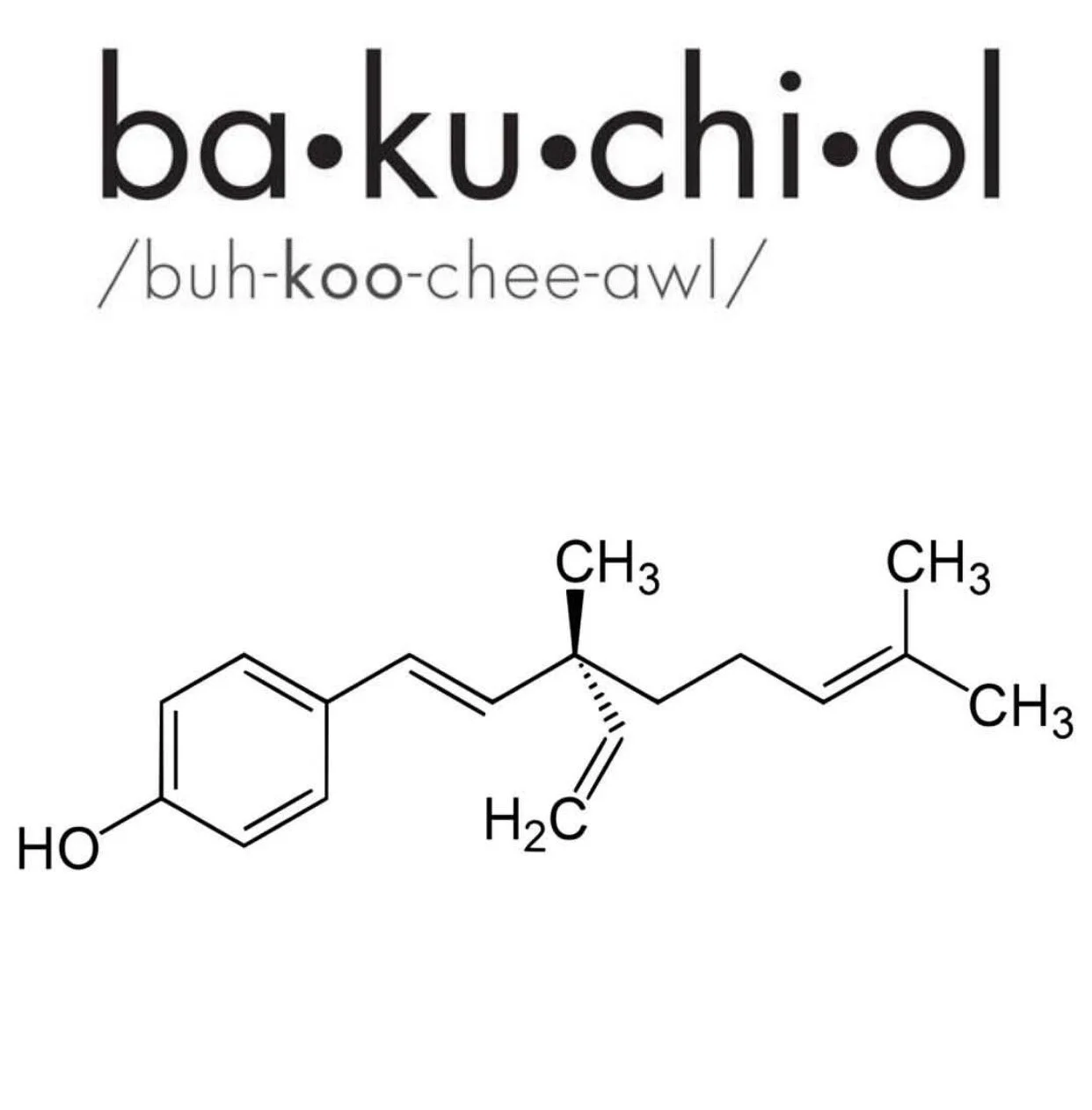
In the ever-evolving industry of skincare, where purveyors of goods are looking to capitalize on the multi-billion dollar anti-aging cosmeceuticals market, we often find ourselves sifting through “buzzwords”, scientific jargon and claims, and new ingredients that, more often than not, come without the crucial scientific evidence needed for proof of efficacy. On occasion, however, as product formulators, we get new raw material ingredients that not only galvanize our industry, but actually have solid evidence-based data to support their usage on skin. Enter Bakuchiol, the plant derived molecule being touted as the true retinol alternative. So, what is this “magical” ingredient, celebrated and glorified across all beauty sectors, all about? Let’s start at the top.
Bakuchiol is an active constituent (meroterpene phenol, in this case) found within the leaves and seeds of the botanical species known as Psoralea corylifolia, or Babchi, as it’s commonly known in the Asian countries where it grows wild. In fact, this plant species has been used for hundreds of years, if not longer, in traditional eastern medicine cultures to treat a variety of systemic and cutaneous ailments. Its therapeutic value is undeniable as it possesses numerous pharmacological properties. So, it ultimately begs the question – is Bakuchiol a viable alternative to Retinol, and can it achieve the same clinical objectives?
Retinol is a natural, alcohol form of Vitamin A in the family of molecules known as retinoids. Other common natural retinoids used in skincare are Retinyl Palmitate (ester form), Retinaldehyde (aldehyde form), and Retinoic Acid (the active acid form), with each maintaining their unique molecular characteristics, pharmacokinetics, and potency. These forms of Vitamin A have been used extensively since the 1940’s to treat a variety of skin conditions, including acne and aging, and they have an abundance of data and statistics proving their therapeutic potential. Vitamin A, and its analogues, are essential cellular “nutrients” that play significant roles in cellular growth and differentiation. In fact, aging can largely be considered a nutritional deficiency, as the suns UV rays rapidly deplete the Vitamin A levels in our skin. They are true anti-aging workhorses! With the evidence clearly in favor of Retinol’s efficacy, what problems would present that would warrant the usage of an alternative?
The unfortunate downstream effects of using retinoids (with some exceptions) can be irritation, excess peeling, dryness, and enhanced sun sensitivity. All too often, retinoids are off the table for sensitive and hyper-reactive skin types. In addition, your stronger pharmaceutical acid forms of Vitamin A can deleteriously impact the integrity of your skin’s protective barrier, leading to a vicious cycle of sub-clinical inflammation which is, by definition, pro-aging.
While a few active raw materials presented to cosmetic formulators have made the bold claim of being retinol alternatives, none of these have come close to the retinol-like characteristics of Bakuchiol. With close scrutiny, let’s analyze the evidence.
While Bakuchiol’s molecular structure is not identical to that of Retinol’s, they do have similar behavioral characteristics. From the perspective of achieving the same clinical outcomes as retinol, without the inherent negatives, Bakuchiol seems to stack up. When we look at an aged face, we see the hallmark signs of lines and wrinkles, mottled pigmentation, loss of elasticity, and an uneven skin tone. The goal of any effective anti-aging ingredient, and something Retinol is known to do well, is reverting these extrinsic symptoms back to a more youthful state. In a double-blind 2018 clinical study published in the British Journal of Dermatology, a comparative analysis was performed, with the participants either using 0.5% Retinol on their face, or 0.5% Bakuchiol. After 12 weeks, measurements were taken to assess each molecules ability to treat these cutaneous signs of aging. Surprisingly, in virtually all measurements taken, both Bakuchiol and Retinol had remarkable clinical efficacy in treating signs of photo-damage, with no statistically significant difference between the two. In layman’s terms, that means they achieved the exact same performance objectives. However, unlike the Retinol group that experienced stinging, erythema, and irritation, the Bakuchiol group presented with none of these clinical adverse effects. (1)
While a visual assessment of efficacy (when using these ingredients, what will consumers SEE when they look in the mirror)
is often considered paramount in clinical studies, alternative types of testing do exist that exploit what an active ingredients true potential is. We know Bakuchiol achieves the same effects as Retinol from a visual and extrinsic perspective, but the real question is how it measures up from a genetic perspective. Not widely used in mass produce commercial skincare, DNA microarrays, or quantitative polymerase chain reaction (qPCR) tests can show us how a single molecule, or group of molecules, influences the genes in our cells. This is the new gold standard in proving an ingredient’s worth – highlighting what genes it turns on and which genes it can turn off. In order to establish Bakuchiol’s true retinol-like characteristics, it was put through this type of rigorous testing and the results were published in a 2014 edition of the International Journal of Cosmetic Science. What did the data show? While Bakuchiol and Retinol do not have the same molecular structure, they do have an almost identical genetic footprint. (2) Modulation of key retinoid-binding and metabolizing genes is Bakuchiol’s strong suit, however, not all retinoid-binding genes were activated in the study. The gene expression of true retinol includes activation of retinoic acid and nuclear receptors, which gives these forms of Vitamin A their inherent toxicity and irritation potential, as well as the strong clinically validated level of efficacy associated with them. Bakuchiol, cleverly, bypassed the expression of these genes in particular. This upregulation (and downregulation) of gene expression, combined with genes associated with key structural proteins such as collagen, elastin, dermo-epidermal junction (DEJ) genes, hyaluronic acid and aquaporin genes, and other essential genes associated with youthful skin, are what makes Bakuchiol a TRUE functional analogue of retinol. When one needs a clear and definitive explanation of an ingredient’s therapeutic potential, you cannot get more concrete evidence than a gene expression analysis. Though not a complete replacement for Vitamin A, an essential nutrient, it’s a frontrunner when you can’t tolerate traditional retinoids.
However, healthy skin is more than just a robust production of skin proteins to reduce lines and wrinkles, as there are many complex molecular and biochemical pathways that contribute to tissue (skin in this case) behaving in a more youthful manner. Bakuchiol has proven to target all main contributing pathways linked to aging from oxidative stress, inflammation, mitochondrial cell energy production, MMP regulation, and protection against UV-induced skin damage. In fact, Bakuchiol outperformed the antioxidant and anti-inflammatory capacity of commodity raw materials known to be gold standards in this category.
Can it get any better? The science suggests it can. Bakuchiol has also shown significant power in treating acneic skin types, not only from its anti-inflammatory capacity, but its ability to regulate sebum output, modify the microbial ecosystem, downregulate localize hormone levels, and reduce hyperkeratinization. (3) And, finally, to further highlight this ingredients protective potential, it has proven to stop the proliferation of cutaneous malignancies such as skin cancer. (4)
While Bakuchiol is a relatively new raw material available to us in skincare, it is an extremely promising active with a wealth of benefits for the skin. As more research is devoted to exploring its therapeutic potential, for now, we can confidently say that, while not identical to Retinol (from a biochemical and gene expression perspective), Bakuchiol has proven to be a worthy competitor, whether used on its own or to potentiate Retinol’s performance when combined. The hype is certainly justified.
(1)
Br J Dermatol. 2018 Jun 27. doi: 10.1111/bjd.16918.
Prospective, randomized, double-blind assessment of topical bakuchiol and retinol for facial photoaging.
(2)
Int J Cosmet Sci. 2014 Jun;36(3):221-30. doi: 10.1111/ics.12117. Epub 2014 Mar 6.
Bakuchiol: a retinol-like functional compound revealed by gene expression profiling and clinically proven to have anti-aging effects.
(3)
RK Chaudhuri & F Marchio, Bakuchiol in the management of acne-affected skin, Cosm & Toil, 126:502-510, 2011
(4)
Oncotarget. 2016 Mar 22; 7(12): 14616–14627.
Bakuchiol suppresses proliferation of skin cancer cells by directly targeting Hck, Blk, and p38 MAP kinase
Regenerative skin therapy: the power of microneedling
When you hear the term “regenerative”, what comes to mind? In biology, the term “regeneration” relates to the ability of a tissue, cell, or organ to recreate or regenerate itself. An entire area of medicine, known as regenerative medicine, focuses on this unique healing potential our bodies maintain. In the world of aesthetics, an industry was formed based on the principle of injuring or wounding the skin, which is degenerative by definition, in order to trick the skin into producing key structural proteins like collagen and elastin. The question remains – is this “destructive” approach to facial rejuvenation the most ideal way to achieve our aesthetic objectives? Is there a better way to stimulate an anti-aging response in the skin that mitigates the excess damage and inflammation caused by these modalities, and taps into a more regenerative model of healing? In fact, there is!
Welcome to the world of microneedling, also known as Collagen Induction Therapy, or CIT. This advanced skin rejuvenation modality, widely used in the dermatology space, has the ability to remodel aged and damaged tissue, all while maintaining skin integrity and defense systems. As sophisticated as this approach to aging skin is, little is still known about its underlying cellular and molecular effects on the architecture of the skin. Microneedling involves the usage of specially designed devices, from automated pen-like tools to manual handheld rollers, that are outfitted with needles of varying lengths, intended to pierce through the top layer of the skin and activate a regenerative response via the wound healing cascade. (1) Unlike the majority of modalities that use thermal energy or mechanical means to completely remove, or ablate, the top layers of the skin, microneedling keeps the epidermis relatively intact in its effort to rejuvenate aged tissue. These unique characteristics of the procedure are what mitigates adverse responses in the skin such as excess inflammation, a hallmark characteristic of traditional wound-based modalities, while potentiating the regenerative capacity the procedure affords us.
In the most basic understanding into mechanism-of-action, the needles (penetrating at different depths in the skin) will pierce the viable cells within our epidermis, initiating not only a wound healing cascade, but also a natural release of skin native protein messenger molecules, known as growth factors and cytokines, among other biological signals. These molecules are what all (nucleated) cells in your body use to literally communicate with one another. Optimizing this cell-to-cell communication is what microneedling does best, in addition to regulating the function of the dominant cell types of the skin including keratinocytes, fibroblasts, and melanocytes, all while respecting their structure in the process. Though the molecular mechanisms of microneedling had not been well understood for some time, histological and gene array studies proved needling causes a significant upregulation of genes associated with tissue regeneration and healing, epithelial proliferation and differentiation, while downregulating a myriad of pro-inflammatory cytokines. (2)
With the understanding that microneedling is a highly efficacious tool for tissue regeneration, it is important to understand treatment parameters and what the goal of the procedure is. It is always vital that you seek out professional training in microneedling, as improper education can lead to inferior clinical results, tissue damage, and potential for infection. In addition, one must always work under their scope of practice as liability can be high.
Historically, the idea was that for microneedling, or any treatment modality for that matter, to be effective, we must create direct damage to the cells in our skin that produce key structural proteins, the Fibroblasts. One of the world’s leading experts in the subject of microneedling, Dr. Lance Setterfield, has shed some insight into the fact that the Dermal-Epidermal Junction (DEJ) is the ideal target site for needling, and not the Fibroblast. He says in his book entitled ‘The Concise Guide to Dermal Needling -3rd Medical Edition’, that “Over the years I have noted clinical improvement in my patients using lesser depths of injury. I began researching the possibilities that would explain the unexpected. Targeting the DEJ corrects loss of structural integrity (the cause of wrinkles and lax skin) through upregulation of genes that result in increased production of collagen.” Essentially, what he discovered was that through targeting the DEJ, the skin was able to effectively increase key structural protein production, and release native signaling molecules that traveled from the epidermis to the dermal fibroblasts coordinating their biological activity. This approach significantly improved clinical outcomes, tapped into a more fetal or embryonic state of regenerative healing, and reduced the downtime often associated with aggressive depths of needling.
So, how does that correlate in a clinical setting? How do we know if we are effectively targeting the DEJ? Histologically, this layer that anchors the epidermis to the dermis presents with a mountainous appearance but does tend to flatten out as we get older. Given this structure, on any given area of the face, the DEJ resides at depths anywhere between 0.4mm to a maximum of 1mm. The most significant clinical results for overall tissue rejuvenation are seen when needling is utilizing depths somewhere in the middle of this range, and depths any deeper are likely to create more collateral damage, inflammation, and less than desirable results.
Recently, the term “Microchanneling” has been used to describe needling the skin at shallow depths, usually in the 0.25mm range. Independent of the marketing term, no matter what depth you are at, if you are using metal needles to pierce the skin, it is and always will be called microneedling. The question remains, does needling at shallow depths provide any benefit? These “cosmetic” depths do have value, since they will still pierce viable epidermal cells and allow them to release epidermal growth factors and cytokines. These will help in stimulating a mild repair and rejuvenation response. At the deeper “medical” depths, you will get a more robust portfolio of regenerative proteins getting produced, and this is important when you are doing more corrective work. Each level of penetration can provide its own unique advantage.
One area of concern I see in the industry is the mindset that these open channels you create with microneedling are an open invitation to allowing any and all products to penetrate better. That is far from true. While yes, needling does allow products to penetrate better at all depths, it is advisable to only use ingredients during microneedling that are considered native to the human body, as foreign material introduced during needling can be presented to the immune system as a dangerous molecule, and granulomas can often form around them. The short list of ingredients advisable to use are sterile saline, pure High Molecular Weigh Hyaluronic Acid (as Low Molecular Weight has proven to be pro-inflammatory and linked to disease (3) (4) (5), and certain concentrated human growth factor products that lean more anti-inflammatory, like the Bone Marrow Stem Cell Growth Factors, versus the pro-inflammatory ones from the Adipose (Fat) Stem Cell Growth Factors. In addition, it is strongly encouraged to avoid anything else other than the above-mentioned molecules, for the first 18-24 hours post needling.
With the right training and education, the right topical adjuvants, staying within your scope of practice, and a smart, common-sense approach to this procedure, microneedling can be a phenomenal tool in your anti-aging and skin regeneration arsenal.
Skin as an immune organ
This is the post excerpt found in the Options tab of Edit Post. Curabitur blandit tempus porttitor. Lorem ipsum dolor sit amet, consectetur adipiscing elit.
It has been well over 40 years since the aesthetic medicine industry, as we know it today, became a unified establishment with a singular purpose: to make the skin look, and hopefully
behave, like its younger self. However, in our beautification efforts, have we inadvertently ignored skin structure and function, and created a model of theoretical cellular regeneration that has no long-term sustainability to the health of our skin? To fully understand the relevance of this concept, it is essential we start at the beginning of a movement whose ideology has remained mired in an antiquated understanding of skin physiology. The general consensus on aging is that, by definition, it is a progressive accumulation of damage at the molecular and cellular level. Barring any chronological and genetic factors, it is extrinsic aging that the movement in question was founded. When the integument is exposed to harsh environmental aggressors, including but not limited to, UV overexposure, chemical toxins, carcinogens and pollutants— a cascade of biological processes are activated that become the catalyst to the acceleration of the aging cell. The mechanism of action pioneered in the late 1960s to rejuvenate classically aged skin was based on the premise that further injury of this damaged tissue, and its subsequent wound healing response, would provide a method of cellular regeneration that, in theory, was therapeutic enough to reward a statistically significant change in its appearance, while being seemingly gentle enough as to not cause major harm.
What I propose is that the gold standard mentioned above, actually does more harm to cutaneous health than good, as it completely disregards the skin’s most basic function, to protect and defend. Since the birth of this aesthetic medicine specialty, our comprehension of skin physiology has evolved at an exponential rate. Given this fact, our approach at treating hallmark photo aging symptoms has yet to catch up. The disconnect between mainstream modalities and providing the skin with what it needs to maintain homeostasis, has never been more heightened. Historically, our skin was categorized as simply being a physical-mechanical barrier between the environment and the body, contributing no other purpose than serving as the body’s proverbial brick wall. And, that two things in particular would turn back the clock on the aging process: removal of the compacted layer of surface corneocytes, thought to be of as non-viable and “dead,” and to directly injure the dermal fibroblast in order to stimulate production of structural protein. We now know this is far from the truth. This misconception has been perpetuated to the point where convoluted marketing practices have undermined empirical data and verified skin science. The skin is the largest organ of the body, and aside from acting as a literal shield of armor, it is a powerful immunologic organ, maintaining and mounting a host defense system when facing foreign threats and unwelcome invasion. In fact, our skin has an estimated 20 billion T cells, outnumbering the amount in the circulatory system, proving that the skin’s number one priority is defense. This, in addition to the billions of commensal microbes that take up residence in our skin, part of the collective microbiome, show that immunosuppression is not the answer to sustainable anti-aging methodologies. Disruption of barrier homeostasis ignites a strong inflammatory and immune suppressive response, that when left in a chronic state, leads to an imbalance in cellular communication, cutaneous microbiome ecology and ultimately cellular senescence. This is the epitome of the pathophysiology behind aging skin. It then begs the question, why is our industry encouraging a cutaneous rejuvenation approach that injures, suppresses and forces the skin to make change, versus supporting and augmenting this immune organ, in our favor, to ignite a regenerative and reparative response, void of inflammation? Inflammation is NOT a prerequisite to repair, as has been proven in the fetal wound-healing model. Direct injury to the fibroblast is NOT required to synthesize new structural protein, as has been proven by cellular cross-talk, with the keratinocyte being the appropriate target for stimulation. And the skin’s barrier has a far more powerful role in dermal health than previously thought, with its integrity being vital to the overall performance, function and homeostasis of the skin as a whole. When we know, definitively, that aging is a direct result of damage, in what context would damaging the skin further produce an opposite effect? The most promising research for cellular regeneration, without insult or injury, leads us to tapping into the skin’s defense system, augmenting and supporting its ability to do what it does best, and maintaining the viability of microbiome symbiosis. The skin and the body are remarkable and fascinating organs that deserve far more respect than they have been given for the latter part of the 20th century. Using targeted skin care technologies that provide a synergistic and coordinated effort, affording us a true partnership with skin function and design, will allow us to finally break free from industry complacency and give us the most sensible and sustainable way to achieve our collective anti-aging objectives.
Stem Cells, Growth Factors & Cytokines in Skincare
This is the post excerpt found in the Options tab of Edit Post. Curabitur blandit tempus porttitor. Lorem ipsum dolor sit amet, consectetur adipiscing elit.
When it comes to skin health, does the “social activity” of your skin cells ever cross your mind? In fact, were you aware that your skin even had a social life? Well-understood biology confirms that your cells have VERY active social interactions going on, and are in constant communication with one another, giving off messages that instruct coordinated biological events, influencing how cells function and behave. All nucleated human cells, outside of red blood cells, communicate with one another by giving off messenger proteins called cytokines and growth factors. These proteins, each one carrying its own unique “instruction”, are widely recognized by the medical literature, including publications such as the Journal of Drugs in Dermatology, as the molecular “keys” that unlock the full potential of your cells to stimulate and optimize skin health. As we begin to grasp the basic fundamentals behind aging, and aging skin in particular, we come to the realization that no matter what research proven ingredient we expose our skin to, if we aren’t optimizing cellular communication, these powerful actives won’t be performing at their highest level. Growth Factors and cytokines allow us to achieve this very objective. However, not all growth factors and cytokines are created equal.
In order to discuss the effects growth factors and cytokines have on the skin, it is necessary to understand it is their combined “pattern” that determines what their effect will be (all cells secrete dozens if not hundreds of growth factors and cytokines.) That pattern is determined by the genetic machinery of the cells being cultured to obtain the growth factors and cytokines - most frequently stem cells from liposuction fat, stem cells from bone marrow, or fibroblasts from male infant foreskins . The role of growth factors and cytokines in wound healing have been studied extensively; they serve multiple functions including regulation of the immune system, promoting collagen synthesis, quenching inflammation, increasing inflammation, reducing scar tissue, and so on.
I want to place emphasis on the ability of these proteins to either INCREASE or DECREASE inflammation. This is their most important aspect when it comes to comparing the different sources of growth factors and cytokines, and ultimately what physiological effect they will have on the skin. It should come as no surprise to anyone that inflammation and aging are synonymous; they are one in the same. Any topical product meant to be anti-aging, in the truest sense of the term, must be strongly anti-inflammatory.
Before I dive into the different sources of these proteins, let’s briefly discuss basic “mechanism of action”. As previously mentioned, these biological signals are how cells “talk” to each other. They work in sort of a “lock and key” fashion. Think of the growth factor or cytokine as the “key”, and the receptor on the surface of the cell as the “lock”. A growth factor (“key”) gets synthesized within a cell; that cell then releases the growth factor where it travels to its neighboring cells and attaches to a receptor on the surface of those cells (“lock”), activating a unique biological response. A cascade of biologic signals gets sent to the heart of the cell, the nucleus, causing a change in genetic expression. The behavior of the neighboring cells is changed. This process continues, to complete the task at hand. This is cell-to-cell communication in its simplest explanation.
Now let’s move on to where these signal proteins are sourced from, and why that is important. The terms growth factor, cytokine and stem cells get thrown around quite often in skin care marketing, with a ton of misinformation being spread around as to what these actually mean. Often it’s the “magical” plant stem cell that gets sensationalized in the skin care arena. Without spending too much time diving into this, when it comes to cell-to-cell communication within HUMAN skin, plants serve no purpose or function. It is physiologically impossible for plant stem cells to communicate with human cells, they just don’t speak the same language. That’s not to say they don’t have some therapeutic value, as research shows potential anti-oxidant capability from certain plant species. That being said, it is vital that we expose the skin to human-derived growth factors and cytokines, in order to achieve the physiological effect we desire.
There are hundreds of different types of growth factors and cytokines. Within the realm of human-derived growth factors, we have multiple cells we can utilize to extract these powerful cellular messenger proteins. Let’s first discuss the process of “extraction”, so you get a better understanding. An easy way to recognize that you have a human derived protein product is when you read the label and it says “_________ conditioned media”, with the blank space being the preferred cell of choice. Independent of the cell used (we will discuss these next), the culturing process is as follows:
-In the lab, the cell of choice is placed into a “glorified” petri dish that contains a media solution. We like to call this a “nutrient-rich broth”. This media is basically a nutritional source for the cells to feed off of and grow.
-As the cells grow, they begin to replicate themselves, exponentially increasing the amount of cells within the culture/media.
-While growing, the cells start to give off/release the growth factors and cytokines that they naturally produce, into the broth/media.
-Once this process is complete, the living cells are filtered out of the broth/media, and what you are left with is the growth factors, cytokines, enzymes, exosomes and other material that the cell released.
-The media is now “conditioned” and ready to be processed and formulated for topical application.
Barring a few additional steps that would be too technical to discuss, this is the process, simplified. Whatever cell is used, each gives off its own unique portfolio of proteins, whose net pattern determines the physiological effect it has on the skin.
There are three main cell types cultured for topical application; Fibroblast cells, Adipose (Fat) Stem Cells and Bone Marrow Stem Cells. The pattern of proteins they produce are vastly different from one another.
Fibroblast Cells: Even though still widely used, this is considered “first generation” growth factor skin care. Fibroblast cells are the dominant cell within connective tissue. These cells are what synthesize key structural protein such as collagen and elastin. When cultured (usually from neonatal tissue), just like any other cell, they begin to release the messenger bio-signals. Fibroblasts, however, are what’s considered “low-ranking” cells. The amount of cytokines and growth factors they release into the media are minute. They are WEAK producers of the bio-signals. With some even teetering on the inflammatory side. Manufacturers will compensate for this by adding a larger dose of the media in their products. This, in no way, improves efficacy, it simply makes the product stink (human protein has a distinct smell!).
Adipose (Fat) Stem Cells: Sourced from middle-aged donors, a byproduct of liposuction waste, they are cheap, easy to culture and grow in abundance. The inherent problem with these cells is that they serve no role in controlled healing within the body. Fat Stem Cells have one fate in life, and that is to create more fat. Fat serves auxillary endocrine function, and when these stem cells are cultured, the portfolio of proteins that get released into the media are strongly PRO-INFLAMMATORY. Metabolically active hormones such as leptin, and cytokines such as TNF-a and IL-6 could disrupt healthy biochemical pathways. Remember that aging and inflammation go hand-in-hand. If the objective of a daily skin care product is to be anti-aging, it is strongly encouraged you avoid the usage of Adipose Stem Cell-derived growth factors and cytokines.
Bone Marrow Stem Cells: This is the master stem cell that controls healing and regeneration throughout the entire body. In fact, it is the only MOBILE stem cell that patrols the body looking for injury. Upon discovery of that injury, it initiates a regenerative response to heal. When cultured (sourced from healthy young adult donors), bone marrow stem cells release an abundance of growth factors and cytokines, whose portfolio is predominantly ANTI-INFLAMMATORY and PRO-HEALING, such as TGF-b3, IGF-1 and IL-10. It is this net-pattern of protein that provides an unparalleled anti-aging response on the skin. No other cell in the body has the ability to fight inflammation quite like this.
It is also important to remember that growth factors do not exist in isolation, and no product should contain a single protein molecule (think the EGF cream craze in the late nineties). You want to use a product that has a physiologically balanced composition of these proteins, and a product whose net-pattern of bio-signals is anti-inflammatory. As an aesthetic practitioner, I encourage you to dive deeper into the research of this technology and constantly ask questions about the products manufacturers are selling you. Armed with this knowledge, you have the power to choose formulations that truly optimize this social network that exists within your skin, and have it work with you, not against you.
Your skin is talking, are you listening?